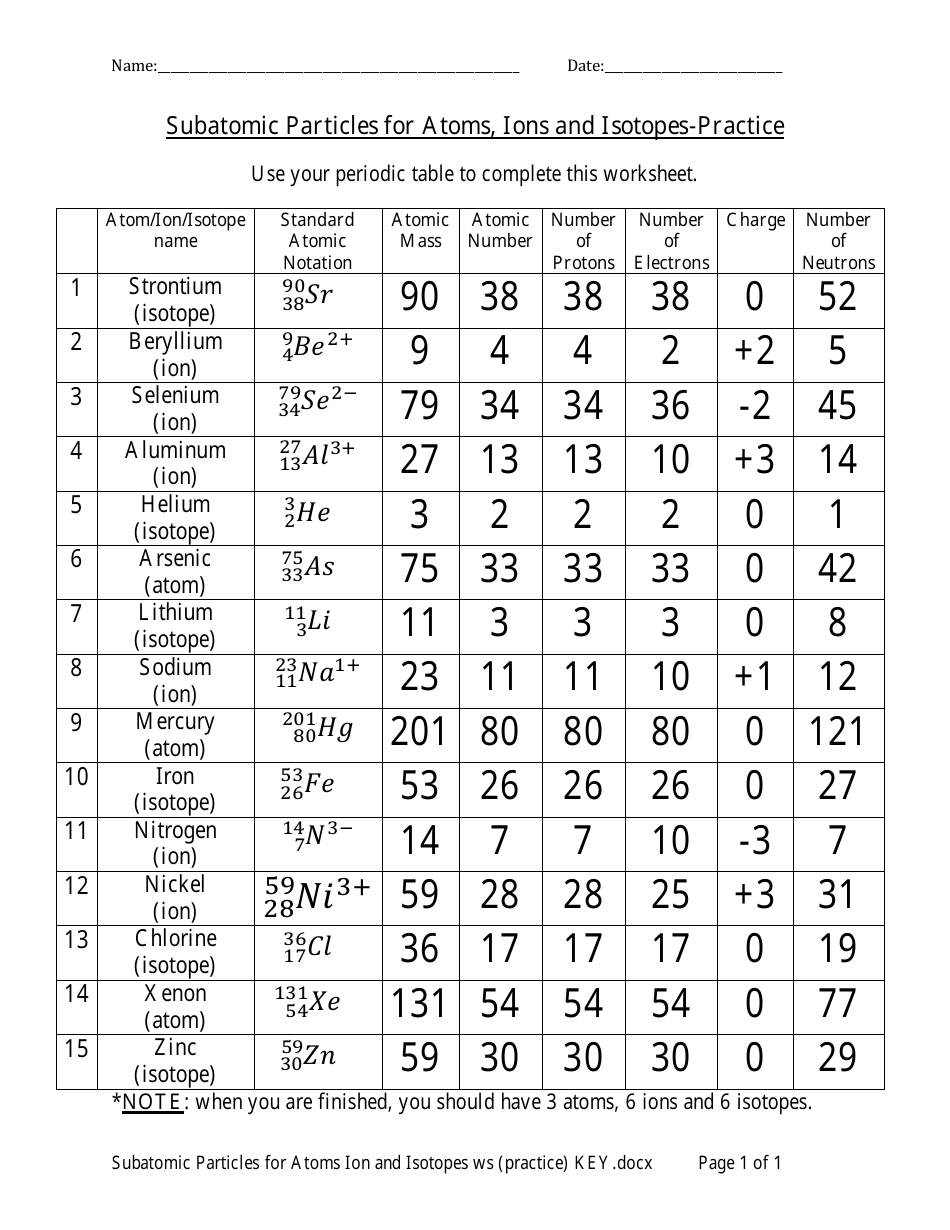
Subatomic Particles for Atoms, Ions and Isotopes Answer Sheet - Mr. Birrell
FAQ
Q: What are subatomic particles?
A: Subatomic particles are the building blocks of atoms.
Q: What are the three main subatomic particles?
A: The three main subatomic particles are protons, neutrons, and electrons.
Q: What is the charge of a proton?
A: A proton has a positive charge.
Q: What is the charge of an electron?
A: An electron has a negative charge.
Q: What is the charge of a neutron?
A: A neutron has no charge.
Q: What is an atom?
A: An atom is the smallest unit of an element that retains its chemical properties.
Q: What is an ion?
A: An ion is an atom or molecule that has gained or lost one or more electrons, resulting in a positive or negative charge.
Q: What is an isotope?
A: An isotope is an atom of the same element that has a different number of neutrons.