Form DBPR-DDC-229 Application for Product Registration - Rx Drugs (Main & Identical) - Florida
What Is Form DBPR-DDC-229?
This is a legal form that was released by the Florida Department of Business & Professional Regulation - a government authority operating within Florida. As of today, no separate filing guidelines for the form are provided by the issuing department.
FAQ
Q: What is DBPR-DDC-229?
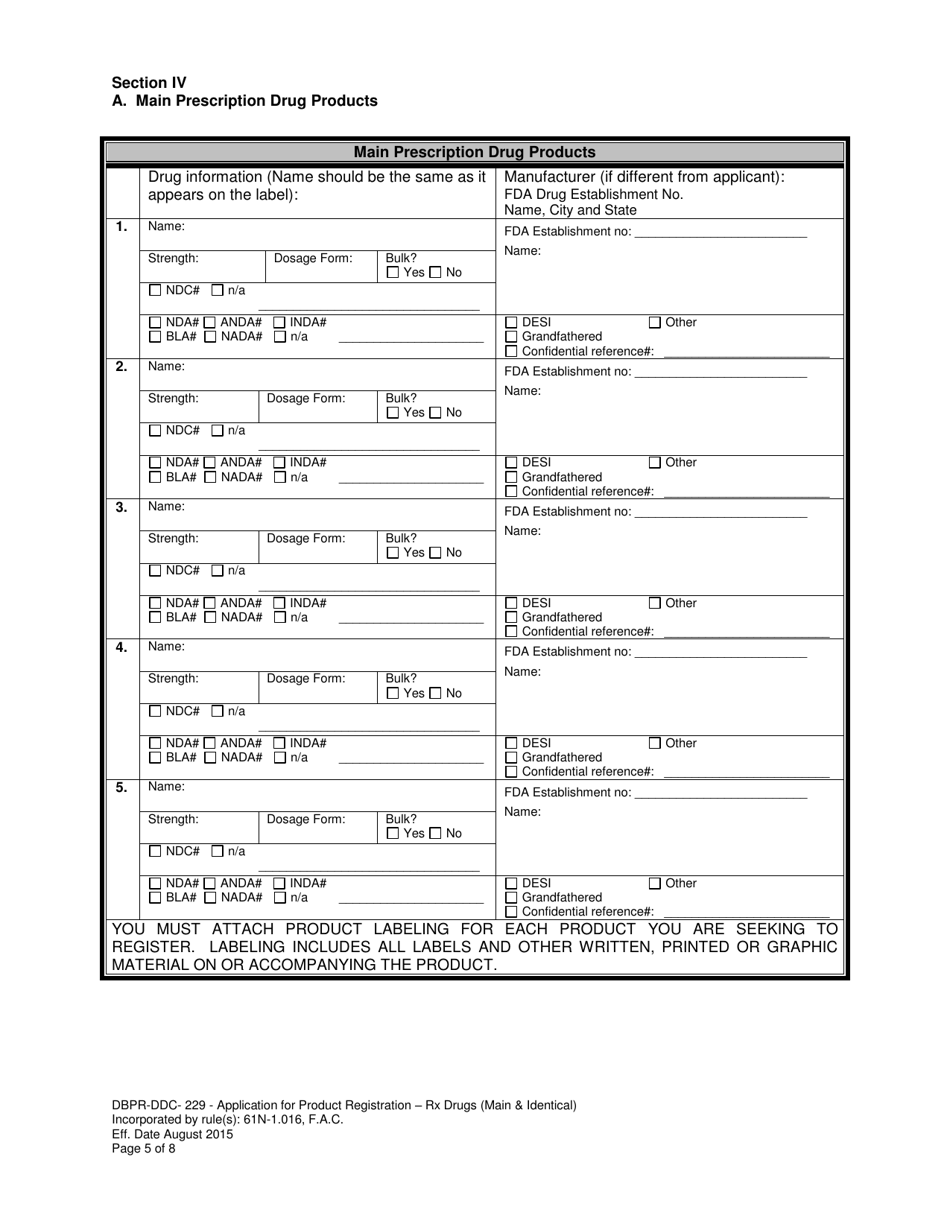
A: DBPR-DDC-229 is an application form for product registration of prescription drugs in Florida.
Q: What does Rx Drugs (Main & Identical) mean?
A: Rx Drugs (Main & Identical) refers to prescription drugs that are the same as the main product being registered.
Q: Who needs to fill out DBPR-DDC-229?
A: Manufacturers or distributors of prescription drugs in Florida need to fill out DBPR-DDC-229.
Q: What information is required in the DBPR-DDC-229 form?
A: The DBPR-DDC-229 form requires information such as the product name, manufacturer information, drug facts, labeling, and packaging information.
Form Details:
- Released on August 1, 2015;
- The latest edition provided by the Florida Department of Business & Professional Regulation;
- Easy to use and ready to print;
- Quick to customize;
- Compatible with most PDF-viewing applications;
- Fill out the form in our online filing application.
Download a printable version of Form DBPR-DDC-229 by clicking the link below or browse more documents and templates provided by the Florida Department of Business & Professional Regulation.