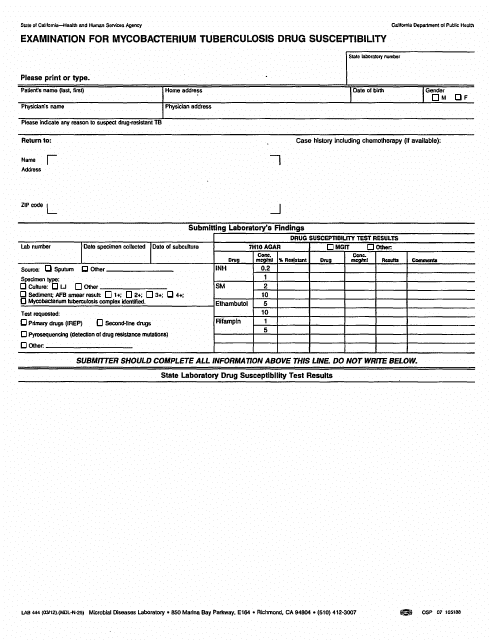
Form LAB444 Examination for Mycobacterium Tuberculosis Drug Susceptibility - California
What Is Form LAB444?
This is a legal form that was released by the California Department of Public Health - a government authority operating within California. As of today, no separate filing guidelines for the form are provided by the issuing department.
FAQ
Q: What is LAB444 examination?
A: The LAB444 examination is a test used for determining drug susceptibility in Mycobacterium tuberculosis.
Q: What is Mycobacterium tuberculosis?
A: Mycobacterium tuberculosis is a bacterium that causes tuberculosis (TB), a contagious infection that primarily affects the lungs.
Q: Why is drug susceptibility testing important for Mycobacterium tuberculosis?
A: Drug susceptibility testing helps determine which drugs will be effective in treating the infection and helps guide appropriate treatment decisions.
Q: What does the LAB444 examination involve?
A: The LAB444 examination involves testing the susceptibility of Mycobacterium tuberculosis to various drugs commonly used to treat TB.
Q: Is the LAB444 examination covered by insurance?
A: Coverage for the LAB444 examination may vary depending on your insurance provider. It is recommended to check with your insurance company for coverage details.
Q: How long does it take to get the results of the LAB444 examination?
A: The time to receive results of the LAB444 examination may vary, but it typically takes several weeks to obtain the final report.
Q: Can the LAB444 examination detect drug-resistant strains of Mycobacterium tuberculosis?
A: Yes, the LAB444 examination is designed to identify drug-resistant strains of Mycobacterium tuberculosis and provides information on which drugs will be effective in treating the infection.
Q: What are the common drugs tested in the LAB444 examination?
A: The LAB444 examination commonly tests the susceptibility of Mycobacterium tuberculosis to drugs such as isoniazid, rifampin, ethambutol, and streptomycin.
Form Details:
- Released on March 1, 2012;
- The latest edition provided by the California Department of Public Health;
- Easy to use and ready to print;
- Quick to customize;
- Compatible with most PDF-viewing applications;
- Fill out the form in our online filing application.
Download a fillable version of Form LAB444 by clicking the link below or browse more documents and templates provided by the California Department of Public Health.