U.S. Department of Health and Human Services Forms
Documents:
429
This document is designed to aid health professionals during in-person consultations for patients using Positive Airway Pressure devices for Obstructive Sleep Apnea. It provides a structured template for capturing vital patient data, diagnosis, and treatment plan.
This document is used for verifying the medical readiness and eligibility of living organ donors before their organ transplant surgery. It ensures that the donor is healthy enough to undergo the procedure and provides necessary information for the transplant team.
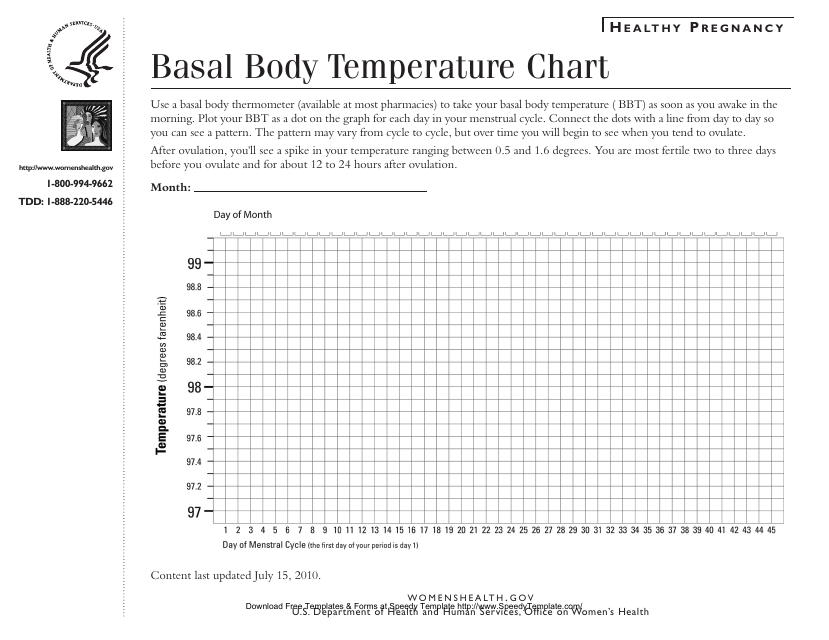
This type of document is used for tracking and recording daily basal body temperature readings, which can help women track their menstrual cycle and fertility.
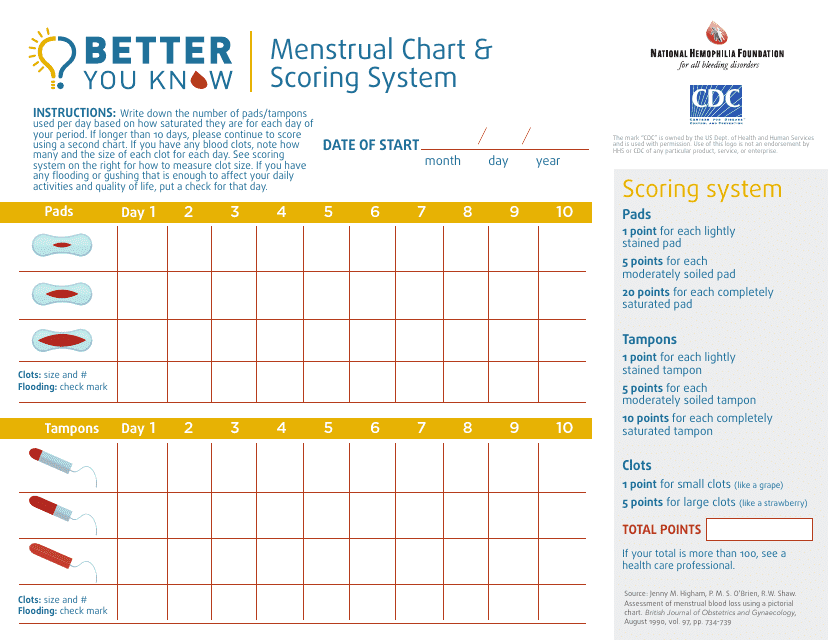
This document is a comprehensive tool utilized to keep detailed track of a woman's menstrual cycle, symptoms and severity. It assists in identifying patterns, health issues and is often used in context of fertility planning or diagnosis of menstrual disorders.
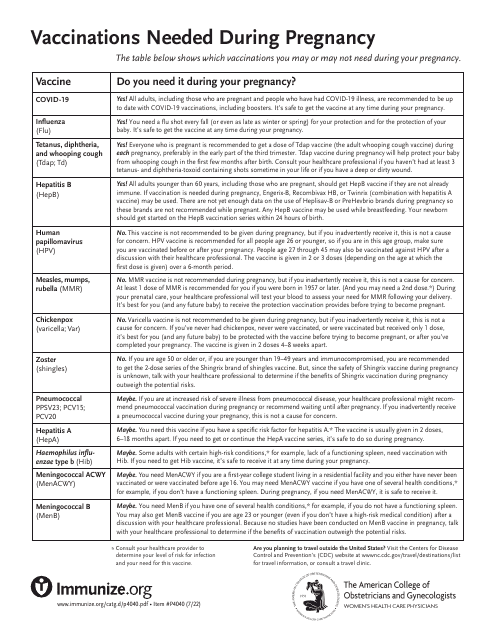
This document provides essential information about the vaccines recommended during pregnancy to safeguard both mother and child from various diseases. It includes details on the timing and safety of each vaccine.
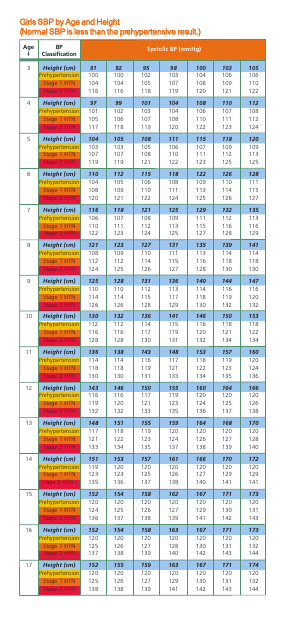
This document is a comprehensive chart providing the standard blood pressure readings for girls, categorized by age and height. It functions as a helpful health assessment tool for paediatric care professionals and caregivers.
This document serves as a reference for healthcare professionals, offering guidance on prescribing opioids for chronic pain management. It aids in assessing patient needs, evaluating their risk of substance misuse, and ensuring ongoing monitoring for safety.
This form is used for tracking the medication intake of Rifampin over a period of 4 months. It is specifically for treating latent tuberculosis infection.
This document outlines the priority categories for the IHS Scholarship Program. It provides information on the eligibility criteria for different priority groups and how applicants can qualify for the scholarship.
This document provides information about the priority categories for the IHS Loan Repayment Program. The program helps healthcare professionals in certain priority areas repay their loans.
This form is used for submitting a request for receivables to the Office of Financial Management (OFM) at the U.S. Food and Drug Administration (FDA).
This Form is used for applying for aid for in-person visitation during the Covid-19 pandemic.
This document provides information on the early warning signs of Autism Spectrum Disorder. It is a valuable resource for parents and caregivers to help identify potential signs of autism in children.
This document is meant to aid and guide in the creation of a comprehensive plan for transitioning patients from a healthcare facility back to their homes or another type of care facility, ensuring that all their care needs will be met upon discharge.
This document provides guidelines and procedures for safely returning a student to school following a concussion. It serves as critical communication between the school, healthcare providers, and parents to ensure a smooth transition and support for the student's health and academic needs.
This document is for keeping track of patient's health progress and details during non-emergency ambulance transportations. It is useful for medical practitioners to communicate the patient's condition and any potential changes during transportation.
This document is a template for recording progress notes related to the use and management of a ventilator. It helps healthcare professionals track and document important information about a patient's respiratory status and treatment progress.
This form is used for filling out a backpack emergency card to provide important information in case of an emergency.
This document provides a template for recording progress notes on vitamin and metabolic assays.
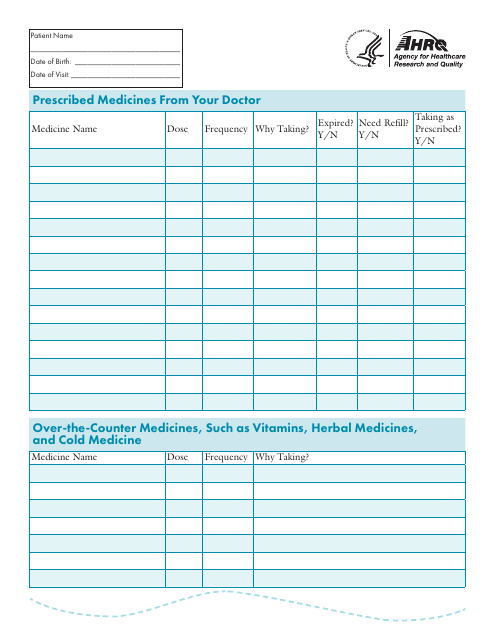
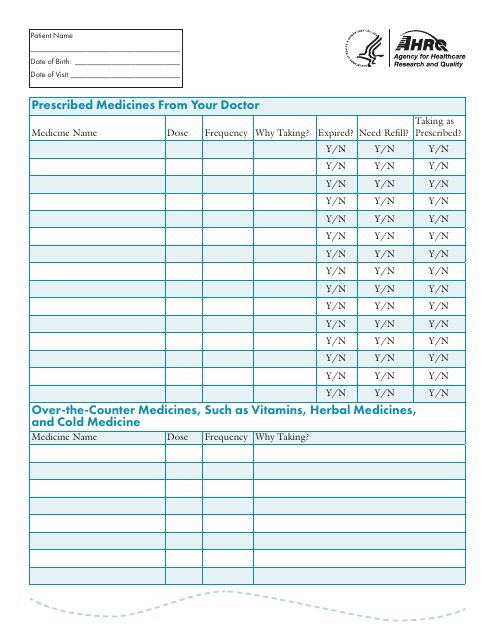
This document is used for keeping a record of the medicines taken by an individual. It helps in keeping track of the medications, their dosages, and the dates they were taken. This can be helpful for managing healthcare and ensuring accurate medication management.
This document outlines the protocols for administering influenza vaccinations. It provides guidelines for healthcare providers to follow in order to ensure the efficient and effective distribution of flu shots.
This document provides a summary of influenza vaccination rates among healthcare personnel. It highlights the percentage of healthcare workers who have received the flu vaccine.
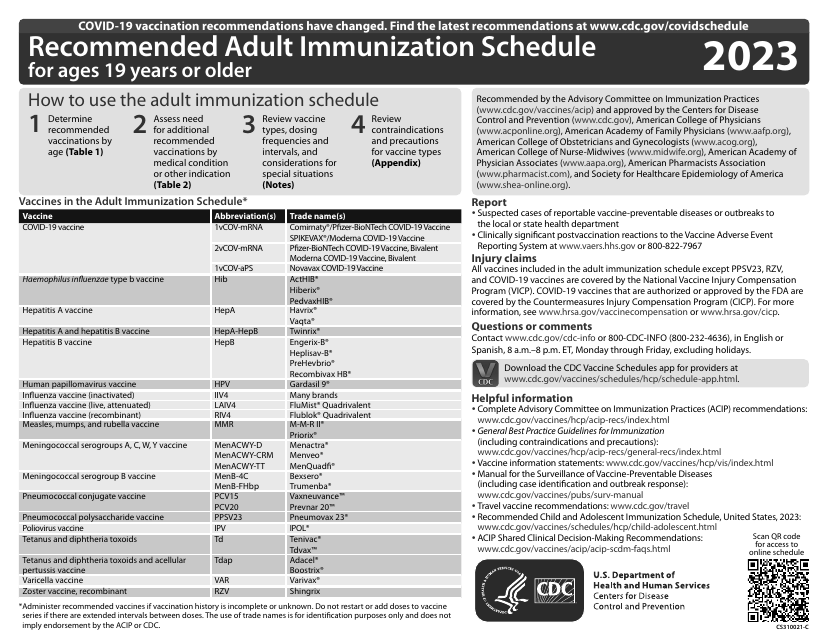
This document provides the CDC's recommended immunization schedule for adults aged 19 years or older. It outlines the vaccines that adults should receive to protect against vaccine-preventable diseases.
This document is a progress note form used for documenting patient progress in a paper format.
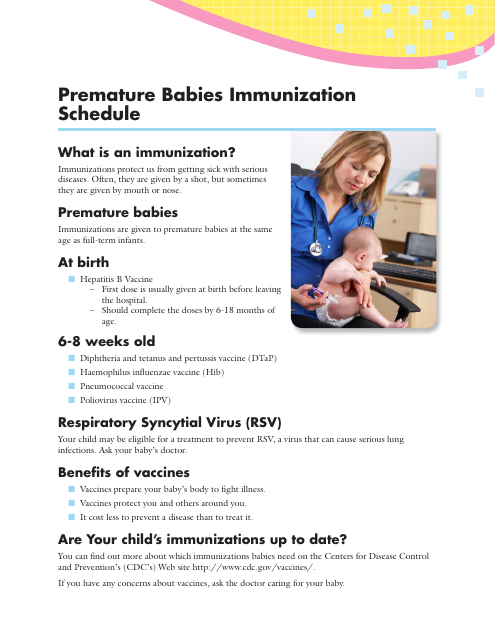
This document outlines the recommended immunization schedule for premature babies. It provides guidance on when and which vaccines should be given to protect these infants from various diseases.
This document is used for keeping track of your personal medicines. It includes the names and dosages of all the medications you take.
This type of document is used to keep track of medications taken, including dosage and frequency. It is helpful for maintaining a record of medications for personal use or for healthcare providers.
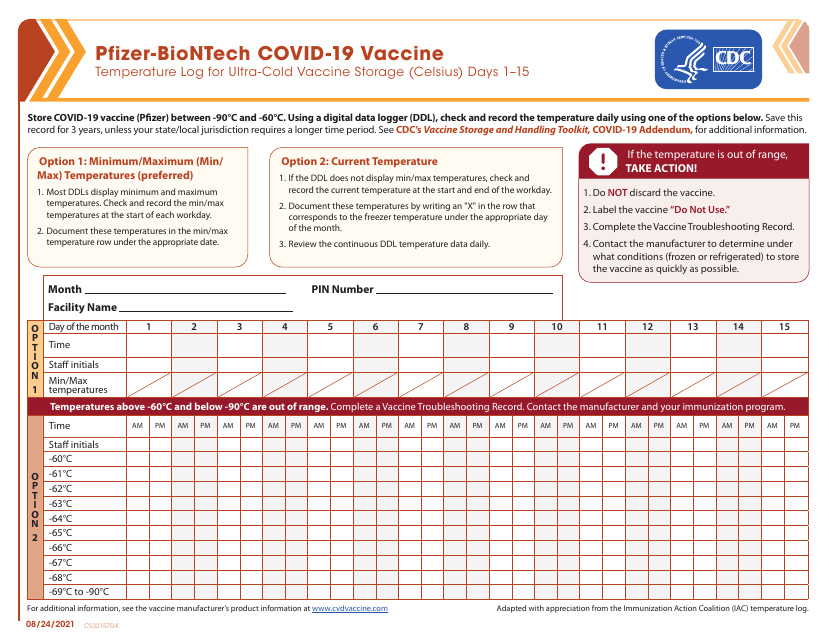
This form is utilized for recording and monitoring the temperature of the Pfizer-Biontech Covid-19 vaccine in ultra-cold storage facilities, with readings in Celsius, thereby ensuring its efficacy during distribution and storage.
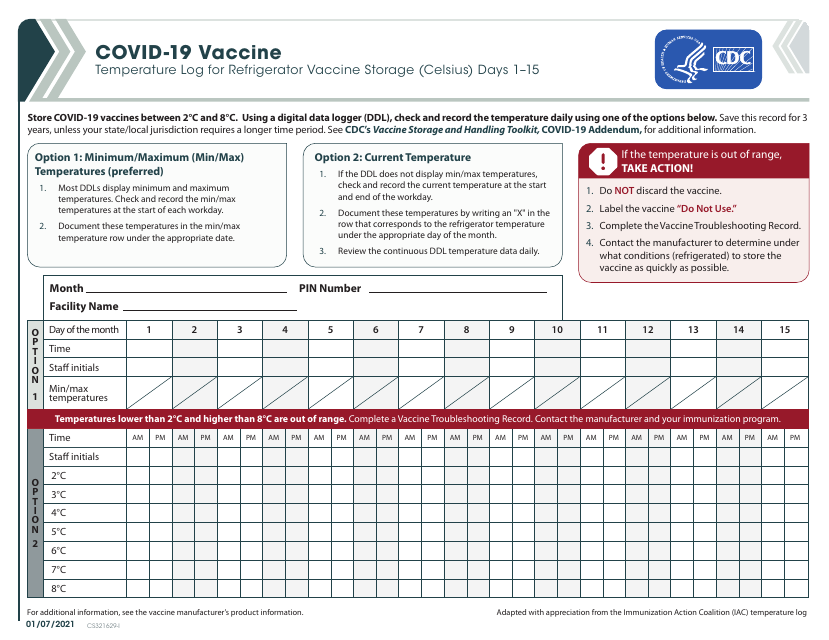
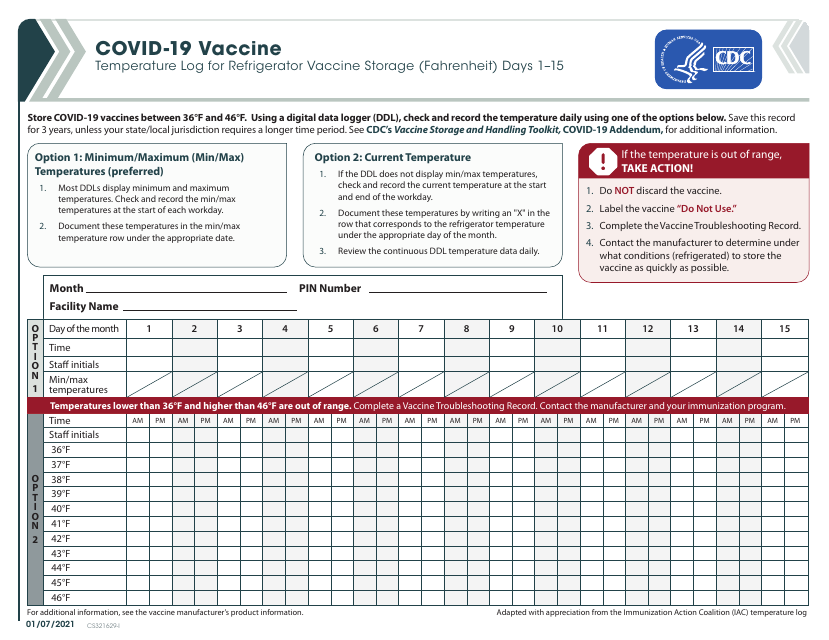
This form is used to record the temperature log for storing Covid-19 vaccines in a refrigerator. It is specifically designed for tracking the temperature of the vaccines stored in Celsius for the first 15 days.
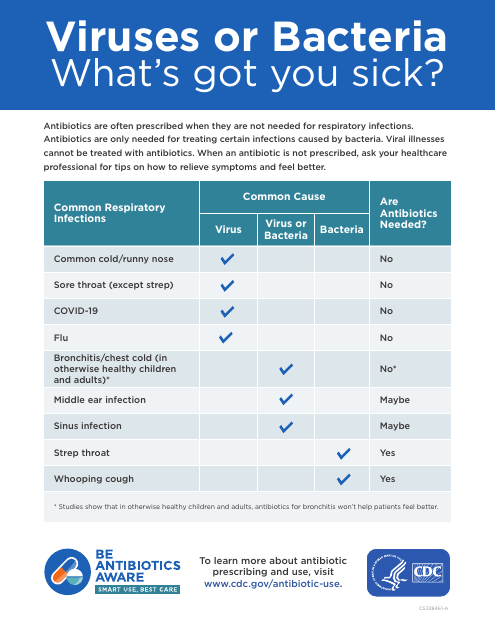
This form is used for reporting whether you have been sick from a virus or bacteria.
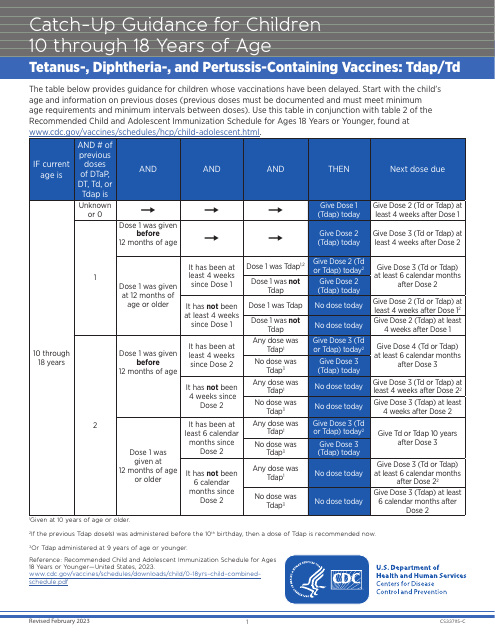
This document provides guidelines for vaccinations and immunizations to ensure health safety of children between ages 10 and 18 who are behind on their vaccination schedules. It outlines the necessary vaccines and their recommended doses based on age and previous vaccination history.
This document is used by healthcare providers to document necessary details about a patient's face-to-face encounter, crucial for determining the requirement and plan for home health services.
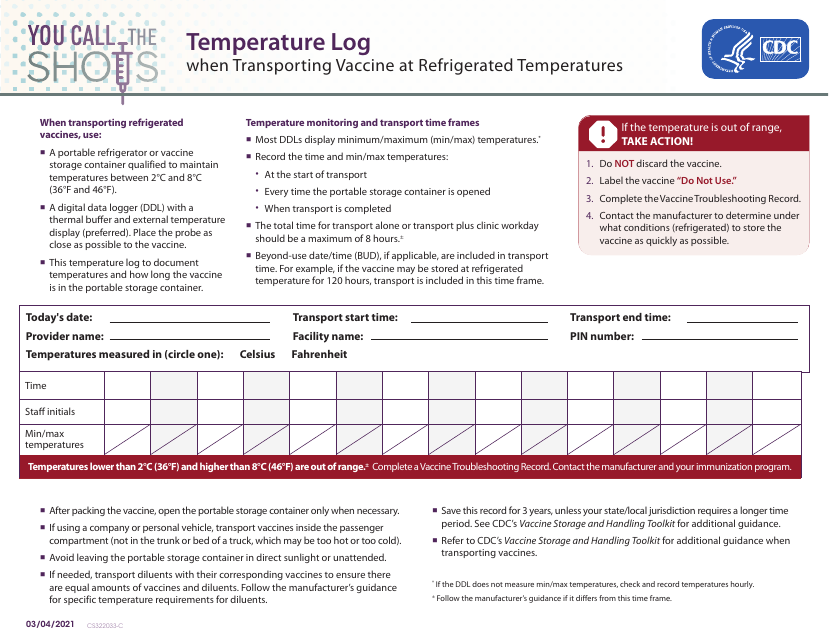
This form is used for meticulously tracking and recording temperatures when vaccines are being transported under refrigerated conditions. It ensures the maintenance of optimal temperature during transit, safeguarding vaccine effectiveness.
This form is used for tracking and recording the temperature of refrigerated Covid-19 vaccines. It provides a systematic way to ensure the vaccines are stored at an optimal temperature within the first 15 days.
This document is for outlining the terms and conditions between a healthcare provider and patient, regarding the use of prescription pain medication. It helps to manage risks and ensure safe, effective treatment.
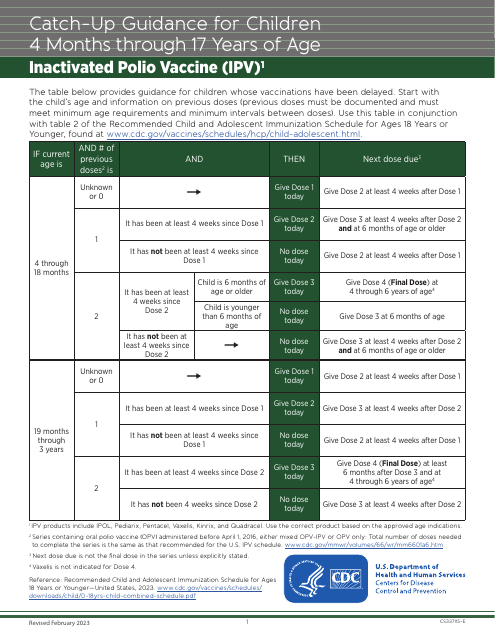
This form provides guidelines for immunization catch-up schedules for children aged 4 months to 17 years within the USA, offering detailed vaccine recommendations and timelines.
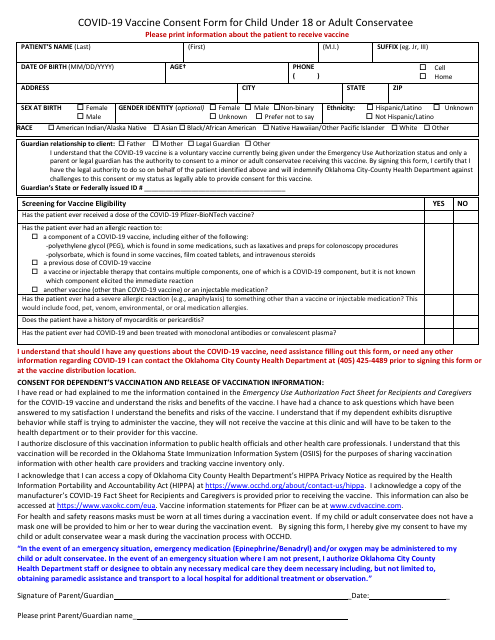
This form is used by legal guardians in Oklahoma to give consent for their child under 18 or adult conservatee to receive the Covid-19 vaccine. It outlines necessary information and guidelines surrounding vaccine administration.
This document is used to conduct an annual survey on patient safety in hospitals. It is an important tool for gathering information and improving patient care.