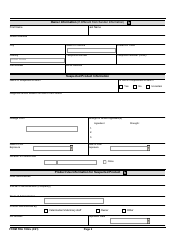
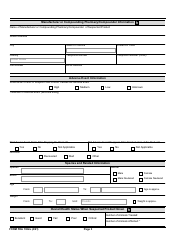
Form FDA1932A Veterinary Adverse Drug Reaction, Lack of Effectiveness, or Product Defect Report (For Voluntary Reporting)
Fill PDF Online
Fill out online for free
without registration or credit card
What Is Form FDA1932A?
This is a legal form that was released by the U.S. Department of Health and Human Services - U.S. Food and Drug Administration on April 1, 2021 and used country-wide. As of today, no separate filing guidelines for the form are provided by the issuing department.
Form Details:
- Released on April 1, 2021;
- The latest available edition released by the U.S. Department of Health and Human Services - U.S. Food and Drug Administration;
- Easy to use and ready to print;
- Yours to fill out and keep for your records;
- Compatible with most PDF-viewing applications;
- Fill out the form in our online filing application.
Download a fillable version of Form FDA1932A by clicking the link below or browse more documents and templates provided by the U.S. Department of Health and Human Services - U.S. Food and Drug Administration.