Drug Safety Templates
At our document knowledge system, we have a comprehensive collection of documents focusing on ensuring the safety of drugs. This collection, also known as drug safety or any derivative name, is curated to provide crucial information and guidelines related to the safe use and monitoring of medications.
Our drug safety documents cover various aspects, including regulatory requirements, reporting forms, comparative guides, and other important resources. For instance, we have a Non-prescription Drug Monograph Attestation Form, specific to Canada, which outlines the necessary attestations for non-prescription drugs in the country. We also offer the Serious Adverse Drug Reaction Reporting Form for Hospitals in Canada, enabling healthcare facilities to report any serious adverse events related to medications.
Our collection extends beyond national boundaries, and we also cater to the drug safety requirements of specific states or regions. The Drug Application document for North Carolina, for example, outlines the application process for obtaining approval to manufacture or distribute drugs in the state.
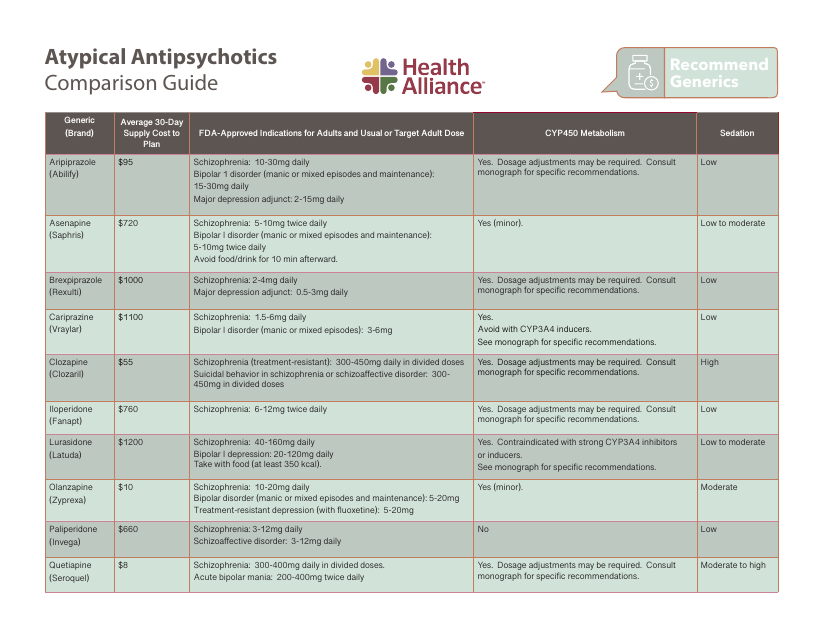
We understand the significance of ensuring adequate information and resources for specific drug classes. Hence, we also offer comprehensive guides such as the Atypical Antipsychotics Comparison Guide from Lexicomp. This guide provides healthcare professionals with a detailed comparison of various atypical antipsychotic medications, assisting them in making informed prescribing decisions.
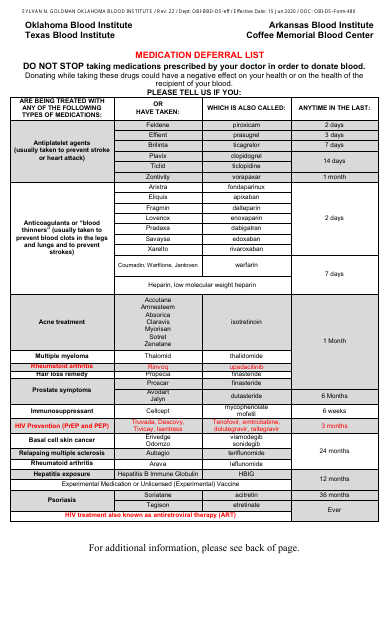
Furthermore, our drug safety collection includes valuable resources like the Medication Deferral List, which provides information on medications that might cause deferral in certain situations or medical conditions. Such resources play a crucial role in preventing adverse drug reactions and improving patient safety.
In summary, our drug safety document collection, including the aforementioned titles, serves as an essential resource for healthcare professionals, researchers, regulatory authorities, and individuals looking to enhance their understanding of drug safety. Whether you are seeking regulatory forms, comparative guides, or informative resources, our collection has got you covered. Find the information you need to ensure safe medication practices and protect patient health.
Documents:
9
This form is used for reporting suspected adverse drug reactions in the state of Connecticut.
This form is used for attesting to the details of a non-prescription drug monograph in Canada.
This form is used for reporting serious adverse drug reactions that occur in hospitals in Canada.
This document is for applying for a Drug Master File (DMF) in Canada. It is used to provide information about the manufacturing, processing, and formulation of a drug product.
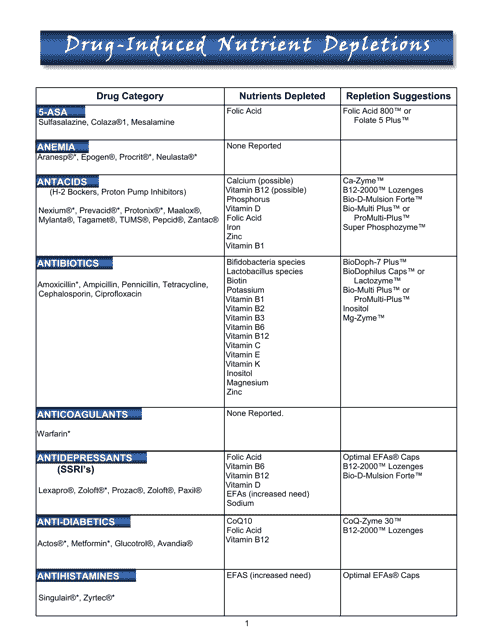
This document provides a chart that lists the nutrient depletions caused by certain medications. It is useful for pharmacists and healthcare professionals to understand the potential dietary changes needed for patients taking these medications.
This guide provides a comparison of different atypical antipsychotic medications, helping users understand their differences and make informed decisions about their use.
This document lists medications that may defer individuals from donating blood. It provides information on which medications can temporarily prevent someone from donating.