FDA Regulations Templates
FDA Regulations
Our webpage is dedicated to providing comprehensive information on FDA regulations, a vital aspect of the healthcare industry. These regulations are put forth by the U.S. Food and Drug Administration (FDA) to ensure the safety, quality, and effectiveness of drugs, biologics, medical devices, food products, dietary supplements, and cosmetics.
At our website, you will find a wealth of resources related to FDA regulations. These resources include guidance documents, application forms, and other tools that help individuals and organizations navigate the regulatory process. Our goal is to empower you with the knowledge and understanding you need to comply with the FDA's requirements and to successfully bring your products to market.
Whether you are a pharmaceutical company seeking approval for a new drug or a manufacturer looking to label your food product, our website provides detailed information on the specific regulations that apply to your industry. We cover a wide range of topics, including the application process, labeling requirements, manufacturing standards, and post-market surveillance.
In addition to providing accurate and up-to-date information, our webpage offers insights and guidance based on industry best practices. We understand that navigating FDA regulations can be complex and overwhelming, but with our user-friendly resources, you can confidently meet the necessary requirements. Our mission is to support businesses in their efforts to bring safe and effective products to consumers while adhering to regulatory standards.
Whether you're an industry professional, a researcher, or a concerned consumer, our webpage is the go-to source for all your FDA regulatory needs. Explore our extensive collection of guidance documents, application forms, and other resources to stay informed and compliant with FDA regulations.
Documents:
8
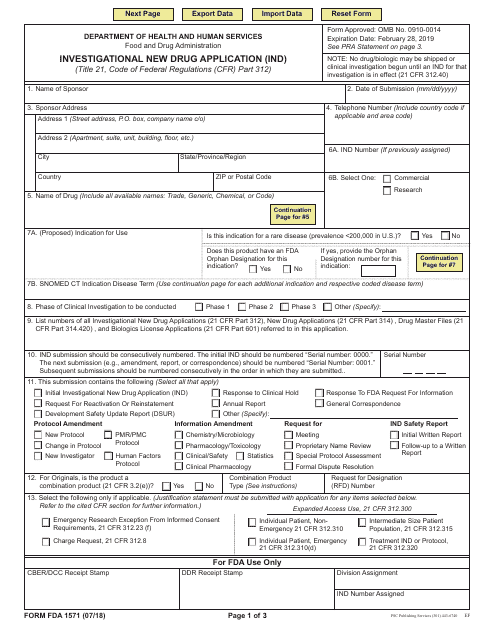
This Form is used for submitting an Investigational New Drug Application (IND) to the FDA. It provides instructions on how to complete the application for conducting clinical trials of a new drug.
This type of document, FDA Form 1571, is used for submitting an Investigational New Drug Application (IND). It is required by the U.S. Food and Drug Administration (FDA) when seeking approval to conduct clinical trials of a new drug.
Formal Meetings Between the FDA and Sponsors or Applicants of Pdufa Products - Guidance for Industry
This Form is used for the approval of food labels in Rhode Island.
This form is used for applying for a variance from the FDA regulations regarding laser light shows, displays, or devices. It allows individuals or organizations to request permission to deviate from the specific requirements outlined in 21 CFR 1040.11(C) in relation to laser safety.